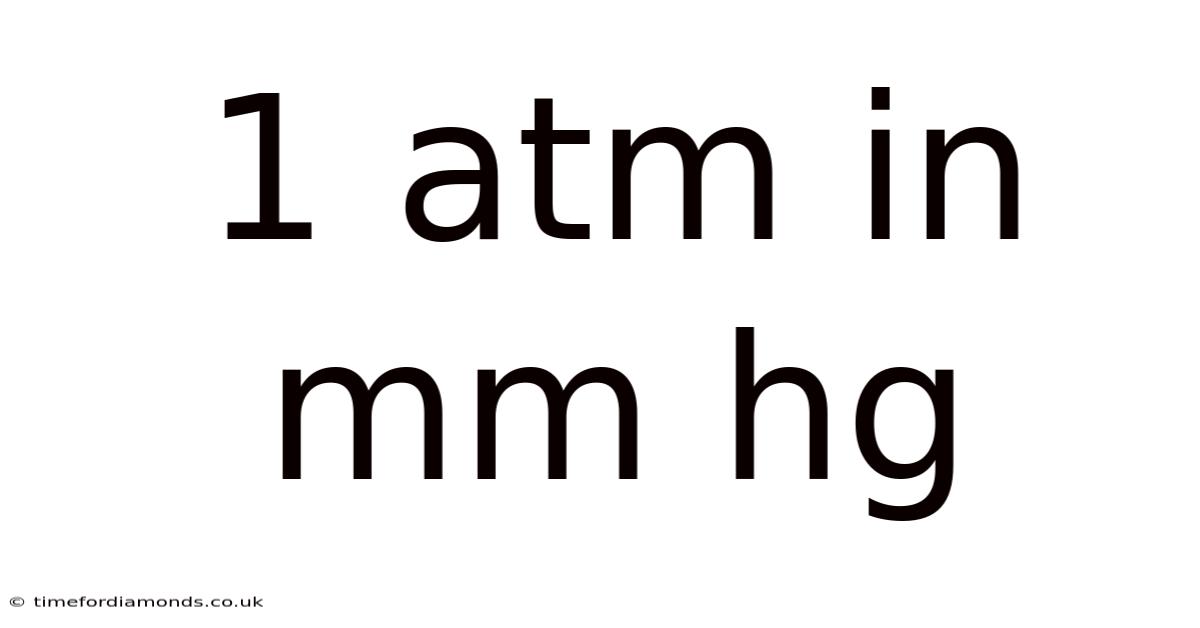Understanding 1 ATM: Pressure, Units, and Applications
Introduction:
This article looks at the meaning and implications of "1 atm," a unit commonly used to express atmospheric pressure. Even so, understanding 1 atm is crucial for anyone studying physics, chemistry, meteorology, or any field involving gases and pressure measurements. We'll explore its definition, conversion to other pressure units like millimeters of mercury (mmHg), the scientific principles behind atmospheric pressure, and its relevance in various scientific fields and everyday life. We will cover the historical context of its measurement, explore real-world applications, and address frequently asked questions.
What is Atmospheric Pressure?
Atmospheric pressure, often represented as P<sub>atm</sub>, is the force exerted by the weight of air molecules in the atmosphere per unit area. This pressure isn't static; it varies with altitude, temperature, and weather conditions. At sea level, the average atmospheric pressure is approximately 1 atm. Now, this pressure is considerable; it’s the force constantly pushing down on us and everything around us. We don't typically notice it because our bodies are adapted to it, and the pressure is roughly equalized inside and outside our bodies Still holds up..
1 ATM: Definition and Equivalents
1 atm (atmosphere) is defined as the average atmospheric pressure at sea level. So while it might seem arbitrary, using 1 atm simplifies calculations and provides a convenient reference point for various applications. It's a unit of pressure, not a physical quantity. That said, you'll want to note that 1 atm is not a fixed constant; it fluctuates slightly due to weather patterns and geographical location.
The standard value for 1 atm is precisely defined in relation to other pressure units, most notably millimeters of mercury (mmHg) and Pascals (Pa).
-
1 atm = 760 mmHg: This equivalence is historically significant. Historically, atmospheric pressure was measured using a barometer, a device that measures pressure by the height of a mercury column. At sea level, the average height of the mercury column is 760 millimeters. This is why 760 mmHg is often used interchangeably with 1 atm Less friction, more output..
-
1 atm = 101325 Pa: Pascals (Pa) are the SI unit of pressure, representing Newtons per square meter (N/m²). The conversion to Pascals provides a more precise and universally understood scientific representation of atmospheric pressure.
Understanding mmHg (Millimeters of Mercury)
The mmHg unit stems from the early methods of measuring atmospheric pressure using a mercury barometer. A higher column indicates higher pressure, and vice versa. In practice, the height of the mercury column in the barometer directly reflects the atmospheric pressure. While mmHg is still used, especially in some medical and scientific contexts, the Pascal (Pa) is the preferred SI unit for pressure But it adds up..
Scientific Principles Behind Atmospheric Pressure
Atmospheric pressure is a direct consequence of gravity acting upon the air molecules above us. Even so, the weight of this column of air exerts a force on the surface below it. At higher altitudes, there's less air above, hence lower atmospheric pressure. This is why climbers experience altitude sickness at high elevations—the reduced pressure affects oxygen uptake.
Several factors influence atmospheric pressure:
-
Altitude: As mentioned above, atmospheric pressure decreases with increasing altitude. This decrease is approximately exponential, meaning the pressure drops more rapidly at lower altitudes than at higher ones.
-
Temperature: Warmer air is less dense than cooler air. That's why, warmer air exerts less pressure for the same volume. Temperature variations contribute to daily and seasonal fluctuations in atmospheric pressure Simple, but easy to overlook..
-
Humidity: Water vapor is lighter than dry air. That's why, humid air exerts slightly lower pressure than dry air at the same temperature and altitude.
-
Weather Systems: High-pressure systems are associated with clear skies and relatively calm weather, while low-pressure systems typically bring stormy weather. These pressure differences drive wind patterns and weather phenomena Easy to understand, harder to ignore..
Real-World Applications of Atmospheric Pressure
Understanding atmospheric pressure is critical in various applications:
-
Meteorology: Weather forecasting heavily relies on accurate pressure readings. Changes in atmospheric pressure are indicators of approaching weather systems, helping meteorologists predict storms, temperature changes, and other weather phenomena Most people skip this — try not to..
-
Aviation: Pilots constantly monitor atmospheric pressure for altitude adjustments and flight safety. Air pressure affects aircraft performance, and accurate pressure readings are vital for navigation and maintaining flight stability Easy to understand, harder to ignore. Still holds up..
-
Medicine: Blood pressure is measured in mmHg, reflecting the pressure of blood against the artery walls. Maintaining appropriate blood pressure is crucial for cardiovascular health. Respiratory therapy also involves monitoring and managing airway pressure.
-
Chemistry: Many chemical processes, especially those involving gases, depend on precise pressure control. In laboratory settings, accurate pressure measurement is essential for conducting experiments and ensuring safe conditions.
-
Industrial Processes: Various industrial processes, such as manufacturing, food preservation (e.g., canning), and chemical engineering, require carefully controlled pressure environments.
-
Diving: Divers need to understand the effects of increasing pressure with depth to avoid decompression sickness ("the bends"). Proper pressure management is vital for deep-sea diving safety.
Calculations and Conversions Involving 1 ATM
Converting between different pressure units often involves simple multiplication or division. The key is knowing the appropriate conversion factors:
-
To convert from atm to mmHg: Multiply the pressure in atm by 760. Take this: 0.5 atm = 0.5 atm * 760 mmHg/atm = 380 mmHg.
-
To convert from atm to Pa: Multiply the pressure in atm by 101325. Take this: 0.5 atm = 0.5 atm * 101325 Pa/atm = 50662.5 Pa.
-
To convert from mmHg to atm: Divide the pressure in mmHg by 760. Take this: 380 mmHg = 380 mmHg / 760 mmHg/atm = 0.5 atm.
-
To convert from Pa to atm: Divide the pressure in Pa by 101325. Take this: 50662.5 Pa = 50662.5 Pa / 101325 Pa/atm = 0.5 atm.
Frequently Asked Questions (FAQ)
-
Q: Is 1 atm always exactly 760 mmHg? A: No, 1 atm is an average value at sea level. Actual atmospheric pressure can vary slightly from 760 mmHg due to weather conditions, altitude, and temperature.
-
Q: Why is the Pascal the preferred unit for pressure? A: The Pascal (Pa) is the SI unit for pressure, providing a consistent and internationally accepted standard for scientific measurements and calculations Turns out it matters..
-
Q: How does atmospheric pressure affect boiling points? A: Lower atmospheric pressure results in lower boiling points. This is why water boils at a lower temperature at higher altitudes.
-
Q: What instruments are used to measure atmospheric pressure? A: Barometers, including aneroid barometers and mercury barometers, are used to measure atmospheric pressure. Digital pressure sensors are also commonly employed.
-
Q: How is atmospheric pressure related to weather forecasting? A: Atmospheric pressure differences are crucial for predicting weather patterns. High-pressure systems are usually associated with fair weather, while low-pressure systems often indicate storms or precipitation Simple, but easy to overlook. Worth knowing..
Conclusion:
Understanding 1 atm – its definition, equivalents in other units like mmHg and Pa, and its influence on various phenomena – is fundamental in numerous scientific disciplines and practical applications. From understanding weather patterns to ensuring safety in aviation and diving, the concept of atmospheric pressure and its measurement are vital. While 760 mmHg provides a historical and convenient reference point, using the SI unit Pascal (Pa) is increasingly preferred for its precision and global standardization. Plus, the importance of accurate pressure measurements cannot be overstated, affecting everything from our daily weather to complex industrial processes. This comprehensive overview aims to equip readers with a thorough understanding of 1 atm and its significance in the world around us Simple, but easy to overlook..